Date: November 18th, 2021
Authors: Kaveh Zakeri, MD, MAS, Nancy Y. Lee, MD
Source: OncLive
The standard of care for patients with locally advanced head and neck squamous cell carcinomas does not substantially differ according to human papillomavirus (HPV) status in the National Comprehensive Cancer Network guidelines.1 Resectable tumors can be treated with surgery followed by adjuvant therapy. Alternatively, definitive chemoradiation therapy with cisplatin is the other dominant treatment paradigm. Incidence of HPV-associated oropharyngeal squamous cell carcinoma has increased rapidly and is associated with higher overall survival (OS) compared with cancers caused by smoking and alcohol.2,3 Given the unique biology of HPV-associated oropharyngeal disease, a separate staging system was developed for these tumors.4
HPV-associated oropharyngeal cancers are more radiosensitive and chemosensitive than cancers caused by smoking and alcohol, yet the traditional treatment paradigms—including high doses of radiation and chemotherapy—were developed prior to the epidemic of HPV-associated disease. De-escalation of therapy has been proposed for HPV-associated oropharyngeal cancer based on data demonstrating high OS and progression-free survival (PFS).5 De-escalation of therapy has been investigated for both definitive surgical and chemoradiation therapy paradigms. Most de-escalated approaches focus on selecting patients according to clinical features, such as disease stage and smoking status, whereas personalized de-escalation reduces treatment intensity for patients according to treatment response.
Transoral Robotic Surgery Followed by Adjuvant Radiotherapy
Transoral robotic surgery (TORS) is a minimally invasive approach that reduces morbidity compared with traditional, open surgery for patients with oropharyngeal cancers. TORS is a standard of care option for patients with resectable tonsil or base of tongue tumors when adequate functional outcome can be preserved. For patients with HPV-associated oropharyngeal disease, postoperative radiation is typically recommended for those with high-risk features including close or positive margins, lymphovascular invasion, perineural invasion, and involved lymph nodes. Adjuvant chemotherapy is recommended for patients with positive margins or lymph nodes with extracapsular extension. Standard doses of radiation consist of 50 to 60 Gy; standard adjuvant chemotherapy typically includes either high-dose or weekly cisplatin.
De-escalated postoperative treatment has been investigated, including reduced intensity of radiation and/or chemotherapy. De-escalation of adjuvant radiation therapy has involved both reduced dose and target volumes. ECOG-ACRIN 3311 (NCT01898494) was a randomized clinical trial in which investigators evaluated reduced dose adjuvant radiation therapy for patients with intermediate postoperative risk factors.6 Patients with low-risk pT1-T2, N0-1 disease with negative margins were observed. Patients with intermediate risk disease (close margins, < 1 mm of extranodal extension, 2 to 4 involved lymph nodes, perineural invasion, or lymphovascular invasion) were randomized to postoperative radiation of either 50 or 60 Gy. High-risk patients with positive margins, greater than 1 mm of extranodal extension, or more than 5 involved nodes received radiation with cisplatin. At a median follow-up of 35.1 months, the 3-year PFS rates were 96.9%, 94.9%, 93.5%, and 90.7% for the low-risk, 50 Gy, 60 Gy, and high-risk arms, respectively.6
Reduced doses of postoperative radiation and chemotherapy were also investigated in the single-arm MC1273 clinical trial (NCT01932697).7 Following surgery, patients received 30 to 36 Gy in 1.5-Gy twice-daily fractions with weekly docetaxel chemotherapy. The rates of 2-year locoregional control, PFS, and OS were 96.2%, 91.1%, and 98.7%, respectively, with 3 years of follow-up. The intensity of 30 to 36 Gy given twice daily compared with the standard 50 to 60 Gy given once daily is unclear.8 Randomized clinical trials are needed to determine whether the MC1273 treatment regimen results in noninferior cure rates with reduced toxicity compared with standard therapy.
The PATHOS trial (NCT02215265) is an ongoing randomized clinical trial investigating a reduction in adjuvant radiation and chemotherapy.9 Patients with low-risk disease are observed postoperatively, patients with intermediate risk factors are randomized to 50 Gy vs 60 Gy, and patients with high-risk features are randomized to 60 Gy alone or 60 Gy with cisplatin.
An additional postoperative de-escalation strategy involves omission of the primary site (tonsil or base of tongue) for patients without primary site risk factors for recurrence (perineural invasion, lymphovascular invasion, or close surgical margins).10 The AVOID trial (NCT02159703) was a single-arm study of adjuvant radiotherapy to the neck alone in patients without primary site risk factors. In total, 60 patients were enrolled and at 2.4 years of median follow-up, only 1 patient had a primary site recurrence. Although this strategy appears promising, more work is necessary to validate these findings and determine the long-term risks and benefits of this approach.
Collectively, these clinical trials demonstrate that TORS followed by adjuvant radiation with or without chemotherapy results in high rates of tumor control. Reduced doses of postoperative radiation and chemotherapy appear promising as a strategy to reduce toxicity while maintaining high rates of cure, and clinical trials investigating these approaches are ongoing.
Two randomized clinical trials have compared TORS plus adjuvant therapy with definitive chemoradiation—the ORATOR trial (NCT01590355) and the ORATOR2 trial (NCT03210103).11,12 The phase 2 ORATOR trial was designed to determine whether TORS would improve 1-year swallowing quality of life. After 25 months of follow-up, patients treated with radiation had a statistically significant improvement in swallowing quality of life, but the difference did not meet the prespecified threshold of clinical significance. There was no difference in PFS or OS between the groups.
The follow-up ORATOR2 study randomized patients to de-escalated radiation-based treatment vs surgery with de-escalated adjuvant therapy.12 The results are pending release. Additional studies are needed to determine the differences between surgery and radiation-based paradigms for treatment of HPV-associated oropharyngeal disease.
Definitive Radiotherapy With Chemotherapy Cetuximab and/or Immunotherapy
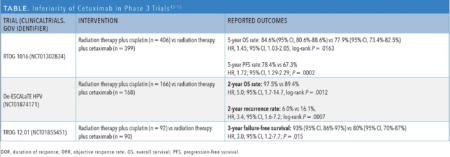
Definitive radiotherapy with high-dose cisplatin is the alternative to TORS-based approaches and is standard for unresectable HPV-associated oropharyngeal tumors. In an attempt to reduce the toxicity of treatment, cetuximab (Erbitux) was proposed as an alternative to cisplatin chemotherapy. Three randomized phase 3 trials compared cisplatin/radiotherapy with cetuximab/radiotherapy: RTOG 1016 (NCT01302834), De-ESCALaTE HPV (NCT01874171), and TROG 12.01 (NCT01855451). Investigators of all 3 trials observed a statistically significant detriment in either PFS or OS for patients treated with cetuximab/radiation (Table).13-15 Additionally, toxicity was not reduced with cetuximab compared with cisplatin.
Omission of systemic therapy with radiation was studied in the randomized phase 2 NRG-HN002 trial (NCT02254278). Patients with p16-positive, nonmetastatic, T1-T2 N1-N2b or T3 N0-N2b (7th edition staging) oropharynx cancer with 10 or fewer pack-years of smoking were randomized to 60 Gy of radiation in 6 weeks with cisplatin vs 60 Gy of radiation in 5 weeks without any systemic therapy. The 2-year PFS rate was 90.5% for cisplatin/radiation vs 87.6% for radiation alone. The radiation alone arm did not meet the prespecified threshold for PFS superiority to 85%. There was no difference in swallowing quality of life between the 2 arms.16
Several randomized trials have investigated the role of immune checkpoint inhibitors (ICIs) in combination with definitive radiation therapy for HPV-associated oropharyngeal cancer. Immunotherapy has been proposed as an alternative to cisplatin for patients with favorable-risk disease and as an additive treatment to cisplatin/radiation for patients with high-risk disease. In patients with favorable-risk disease, the goal of replacing cisplatin with immunotherapy is to reduce the toxicity of treatment while maintaining high cure rates. Investigators are testing this strategy (NRG-HN005, CCTG HN.9 [NCT03410615], and KEYCHAIN [NCT03383094]) and have incorporated ICIs such as nivolumab (Opdivo), durvalumab (Imfinzi), tremelimumab, and pembrolizumab (Keytruda) into the radiation-based treatment paradigm.17-19
For patients with high-risk disease, ICIs have been added to the cisplatin/radiation backbone. Investigators of the randomized phase 3 JAVELIN HEAD AND NECK 100 trial (NCT02952586) evaluated the addition of avelumab (Bavencio) to cisplatin/radiotherapy and included patients with high-risk HPV-associated oropharynx cancer. There was no improvement in PFS with the addition of avelumab to cisplatin/radiotherapy.20 Additionally, investigators of the randomized phase 3 Groupe Oncologie Radiotherapie Tete et Cou REACH trial (NCT02999087) did not observe a benefit with radiation/avelumab/ cetuximab vs cisplatin/radiation.21
These randomized clinical trials highlight the importance of radiosensitizing cisplatin in combination with radiation therapy. Replacement of cisplatin with cetuximab and omission of cisplatin led to inferior tumor control without improvements in toxicity. For patients with high-risk disease, immunotherapy has not demonstrated a benefit when added to cisplatin/radiation or radiation/cetuximab. For patients with favorable-risk disease, ongoing clinical trials will determine whether there is a role for radiation/ immunotherapy.
Personalized Chemoradiation Therapy Based on Hypoxia Imaging
Personalized treatment strategies involve tailoring the intensity of radiation and chemotherapy to individual patient biology and tumor response. Among patients with HPV-associated oropharyngeal cancer, there is heterogeneity in tumor biology and resistance to treatment.22-24 Nonpersonalized treatment paradigms including omission of systemic therapy and replacement of cisplatin with cetuximab were unsuccessful, potentially because differences in tumor biology were not accounted for. Personalized treatment according to tumor biology may facilitate successful tailoring of treatment intensity. Tumor hypoxia is associated with radioresistance and inferior locoregional control and OS in head and neck cancer.25,26
18F-fluoromisonidazole (18F-FMISO) positron emission tomography (PET) is a novel method of measuring tumor hypoxia in vivo and can predict treatment outcomes in patients with head and neck cancer.27 De-escalation of radiation and chemotherapy based on tumor hypoxia using 18F-FMISO PET is a promising treatment strategy for HPV-associated oropharynx cancer. An initial pilot study at Memorial Sloan Kettering Cancer Center in New York, New York, demonstrated successful de-escalation to 60 Gy of radiation with cisplatin for patients with resolution of hypoxia according to 18F-FMISO PET imaging.28 Among the 33 enrolled patients, the 2-year locoregional control and OS rates were 100%.
The subsequent 30 ROC trial (NCT03323463) also conducted by investigators at Memorial Sloan Kettering Cancer Center investigated whether a dramatic reduction in radiation dose to 30 Gy with high-dose cisplatin or carboplatin with 5-fluorouracil could result in successful de-escalation. Patients had resection of the primary tumor prior to chemoradiation and planned neck dissection at 4 months post chemoradiation to measure pathologic response. Patients with resolution of hypoxia on 18F-FMISO PET imaging were treated to 30 Gy with 2 cycles of chemotherapy. Among the 19 enrolled patients, the 2-year rates of locoregional control and OS were 94.4% and 94.7%, respectively.29
Phase 2 of the 30 ROC trial examined the same strategy of resection of the primary tumor followed by 30 Gy of radiation and 2 cycles of chemotherapy with omission of planned neck dissections. The rates of 1-year locoregional control, distant metastasis-free survival, and OS were 94%, 100%, and 100%, respectively, at median follow-up of 1 year.30 All 8 reported local recurrences were in the neck and successfully salvaged with surgery. A subsequent phase of the 30 ROC trial is ongoing with omission of both resection of the primary tumor and planned neck dissection.
Next Steps in the Field
The standard-of-care treatment paradigms for patients with locally advanced HPV-associated head and neck cancer include up-front surgery followed by adjuvant radiation with or without chemotherapy and definitive radiation with cisplatin. De-escalation of postoperative adjuvant therapy is the subject of investigation. Replacement of cisplatin with cetuximab or omission of cisplatin with definitive radiotherapy have not been successful. The addition of immunotherapy to definitive radiation-based treatment has not demonstrated a benefit thus far. A personalized treatment paradigm based on hypoxia imaging is promising and may yield a successful, personalized de-escalation strategy for patients with HPV-associated oropharynx cancer.
Kaveh Zakeri, MD, MAS, is a radiation oncologist at Memorial Sloan Kettering Cancer Center in New York, New York.
Nancy Y. Lee, MD, is vice chair of the Department of Radiation Oncology, service chief of head and neck radiation oncology, and service chief of proton therapy at Memorial Sloan Kettering Cancer Center in New York, New York.
References
- NCCN. Clinical Practice Guidelines in Oncology. Head and neck cancers, version 3.2021. Accessed October 29, 2021. https://www.nccn.org/professionals/physician_gls/ pdf/head-and-neck.pdf
- Ang KK, Harris J, Wheeler R, et al. Human papillomavirus and survival of patients with oropharyngeal cancer. N Engl J Med. 2010;363(1):24-35. doi:10.1056/NEJMoa0912217
- Chaturvedi AK, Engels EA, Pfeiffer RM, et al. Human papillomavirus and rising oropharyngeal cancer incidence in the United States. J Clin Oncol. 2011;29(32):4294- 4301. doi:10.1200/JCO.2011.36.4596
- O’Sullivan B, Huang SH, Su J, et al. Development and validation of a staging system for HPV-related oropharyngeal cancer by the International Collaboration on Oropharyngeal cancer Network for Staging (ICON-S): a multicentre cohort study. Lancet Oncol. 2016;17(4):440-451. doi:10.1016/S1470-2045(15)00560-4
- Adelstein DJ, Ismaila N, Ku JA, et al. Role of treatment deintensification in the management of p16+ oropharyngeal cancer: ASCO provisional clinical opinion. J Clin Oncol. 2019;37(18):1578-1589. doi:10.1200/JCO.19.00441
- Ferris RL, Flamand Y, Weinstein GS, et al. Updated report of a phase II randomized trial of transoral surgical resection followed by low-dose or standard postoperative therapy in resectable p16+ locally advanced oropharynx cancer: a trial of the ECOG-ACRIN cancer research group (E3311). J Clin Oncol. 2021;39(suppl 15):6010. doi:10.1200/ JCO.2021.39.15_suppl.6010
- Ma DJ, Price KA, Moore EJ, et al. Phase II evaluation of aggressive dose de-escalation for adjuvant chemoradiotherapy in human papillomavirus-associated oropharynx squamous cell carcinoma. J Clin Oncol. 2019;37(22):1909-1918. doi:10.1200/ JCO.19.00463
- Garden AS. Not all 30-Gy regimens are equal. J Clin Oncol. 2019;37(36):3558-3559. doi:10.1200/JCO.19.01666
- Owadally W, Hurt C, Timmins H, et al. PATHOS: a phase II/III trial of risk-stratified, reduced intensity adjuvant treatment in patients undergoing transoral surgery for human papillomavirus (HPV) positive oropharyngeal cancer. BMC Cancer. 2015;15:602. doi:10.1186/s12885-015-1598-x
- Swisher-McClure S, Lukens JN, Aggarwal C, et al. A phase 2 trial of alternative volumes of oropharyngeal irradiation for de-intensification (AVOID): omission of the resected primary tumor bed after transoral robotic surgery for human papilloma virus–related squamous cell carcinoma of the oropharynx. Int J Radiat Oncol Biol Phys. 2020;106(4):725-732. doi:10.1016/j.ijrobp.2019.11.021
- Nichols AC, Theurer J, Prisman E, et al. Radiotherapy versus transoral robotic surgery and neck dissection for oropharyngeal squamous cell carcinoma (ORATOR): an open-label, phase 2, randomised trial. Lancet Oncol. 2019;20(10):1349-1359. Published correction appears in Lancet Oncol. 2019;20(12):e663.
- Nichols AC, Lang P, Prisman E, et al. Treatment de-escalation for HPV-associated oropharyngeal squamous cell carcinoma with radiotherapy vs. trans-oral surgery (ORATOR2): study protocol for a randomized phase II trial. BMC Cancer. 2020;20(1):125. doi:10.1186/s12885-020-6607-z
- Gillison ML, Trotti AM, Harris J, et al. Radiotherapy plus cetuximab or cisplatin in human papillomavirus-positive oropharyngeal cancer (NRG Oncology RTOG 1016): a randomised, multicentre, non-inferiority trial. Lancet. 2019;393(10166):40-50. Published correction appears in Lancet. 2020;395(10226):784.
- Mehanna H, Robinson M, Hartley A, et al; De-ESCALaTE HPV Trial Group. Radiotherapy plus cisplatin or cetuximab in low-risk human papillomavirus-positive oropharyngeal cancer (De-ESCALaTE HPV): an open-label randomised controlled phase 3 trial. Lancet. 2019;393(10166):51-60. doi:10.1016/S0140-6736(18)32752-1
- Rischin D, King M, Kenny L, et al. Randomized trial of radiotherapy with weekly cisplatin or cetuximab in low-risk HPV-associated oropharyngeal cancer (TROG 12.01) – a Trans-Tasman Radiation Oncology Group study. Int J Radiat Oncol Biol Phys. 2021;111(4):876-886. doi:10.1016/j.ijrobp.2021.04.015
- Yom SS, Torres-Saavedra P, Caudell JJ, et al. Reduced-dose radiation therapy for HPV-associated oropharyngeal carcinoma (NRG Oncology HN002). J Clin Oncol. 2021;39(9):956-965. doi:10.1200/JCO.20.03128
- Spreafico A, Sultanem K, Chen B, et al. A randomized phase II study of cisplatin plus radiotherapy versus durvalumab plus radiotherapy followed by adjuvant durvalumab versus durvalumab plus radiotherapy followed by adjuvant tremelimumab and durvalumab in intermediate risk, HPV-positive, locoregionally advanced oropharyngeal squamous cell cancer (LA-OSCC) (Canadian Cancer Trials Group HN.9). Ann Oncol. 2018;29(suppl_8):VIII399. doi:10.1093/annonc/mdy287.080
- De-intensified radiation therapy with chemotherapy (cisplatin) or immunotherapy (nivolumab) in treating patients with early-stage, HPV-positive, non-smoking associated oropharyngeal cancer. ClinicalTrials.gov. Updated October 26, 2021. Accessed October 29, 2021. https://clinicaltrials.gov/ct2/show/NCT03952585
- Chemoradiation vs immunotherapy and radiation for head and neck cancer. ClinicalTrials.gov. Updated October 8, 2021. Accessed October 29, 2021. https://clinicaltrials. gov/ct2/show/NCT03383094
- Lee NY, Ferris RL, Psyrri A, et al. Avelumab plus standard-of-care chemoradiotherapy versus chemoradiotherapy alone in patients with locally advanced squamous cell carcinoma of the head and neck: a randomised, double-blind, placebo-controlled, multicentre, phase 3 trial. Lancet Oncol. 2021;22(4):450-462. doi:10.1016/S1470- 2045(20)30737-3
- Bourhis J, Tao Y, Sun X, et al. Avelumab-cetuximab-radiotherapy versus standards of care in patients with locally advanced squamous cell carcinoma of head and neck (LA-SCCHN): randomized phase III GORTEC-REACH trial. Ann Oncol. 2021;32(suppl 5):S1283-S1346. doi:10.1016/annonc/annonc741
- Kimple RJ, Smith MA, Blitzer GC, et al. Enhanced radiation sensitivity in HPV-positive head and neck cancer. Cancer Res. 2013;73(15):4791-4800. doi:10.1158/0008-5472.CAN-13-0587
- Vainshtein J, McHugh JB, Spector ME, et al. Human papillomavirus-related oropharyngeal cancer: HPV and p16 status in the recurrent versus parent tumor. Head Neck. 2015;37(1):8-11. doi:10.1002/hed.23548
- Rieckmann T, Tribius S, Grob TJ, et al. HNSCC cell lines positive for HPV and p16 possess higher cellular radiosensitivity due to an impaired DSB repair capacity. Radiother Oncol. 2013;107(2):242-246. doi:10.1016/j.radonc.2013.03.013
- Terris DJ. Head and neck cancer: the importance of oxygen. Laryngoscope. 2000;110(5 Pt 1):697-707. doi:10.1097/00005537-200005000-00001
- Brizel DM, Dodge RK, Clough RW, Dewhirst MW. Oxygenation of head and neck cancer: changes during radiotherapy and impact on treatment outcome. Radiother Oncol. 1999;53(2):113-117. doi:10.1016/s0167-8140(99)00102-4
- Rasey JS, Koh WJ, Evans ML, et al. Quantifying regional hypoxia in human tumors with positron emission tomography of [18F]fluoromisonidazole: a pretherapy study of 37 patients. Int J Radiat Oncol Biol Phys. 1996;36(2):417-428. doi:10.1016/s0360-3016(96)00325-2
- Lee N, Schoder H, Beattie B, et al. Strategy of using intratreatment hypoxia imaging to selectively and safely guide radiation dose de-escalation concurrent with chemotherapy for locoregionally advanced human papillomavirus-related oropharyngeal carcinoma. Int J Radiat Oncol Biol Phys. 2016;96(1):9-17. doi:10.1016/j.ijrobp.2016.04.027
- Riaz N, Sherman E, Pei X, et al. Precision radiotherapy: reduction in radiation for oropharyngeal cancer in the 30 ROC trial. J Natl Cancer Inst. 2021;113(6):742-751. doi:10.1093/jnci/djaa184
- Lee NY, Sherman EJ, Schöder H, et al. The 30 ROC trial: precision intra-treatment imaging guiding major radiation reduction in human papillomavirus related oropharyngeal cancer. J Clin Oncol. 2021;39(suppl 15):6019. doi:10.1200/JCO.2021.39.15_suppl.6019

Leave A Comment
You must be logged in to post a comment.