Source: www.clinicaladvisor.com
Authors: Christina Alvarez, PA-C, Corinne I. Alois, MS, PA-C, Louise Lee, EdD, MHA, PA-C
Oral leukoplakia is a common premalignant condition most commonly identified in the smoking patient population. Once oral leukoplakia is diagnosed, the fundamental goal is to minimize progression towards invasive carcinoma through the use of various treatment methods such as traditional scalpel excision, cryotherapy, and carbon dioxide (CO2) laser therapy.
Epidemiology and Pathogenesis of Oral Leukoplakia
Leukoplakia is defined as a white, painless, plaque most commonly occurring on the buccal mucosa, lateral tongue, or floor of the mouth.1 Leukoplakia is considered one of the most common oral potentially malignant disorders (OPMDs) with an estimated worldwide prevalence of 1.5% to 2.6%.1 This premalignant condition, which can lead to oral cancer, particularly squamous cell carcinoma, presents with various transformation rates ranging from 0.1% to 17.5%.2 Due to variability in transformation to malignancy, identification, treatment, and monitoring is imperative.
The etiology of oral leukoplakia is multifactorial and many cases are considered idiopathic; however, several strong risk factors are associated with this condition, the most common being chronic exposure to all forms of tobacco products, which cause mucosal irritation over time.1,2 Prolonged use of tobacco products is attributed to the conversion of normal cells to hyperplasia, dysplasia, and eventually carcinoma in situ or invasive carcinoma. It is reported that leukoplakia is 6 times more common in smokers than nonsmokers.1
Oral leukoplakia is most commonly seen in men aged 40 years and older, particularly after years of chronic tobacco use.1,2 Other common contributory risk factors include: alcohol use, betel nut use, genetics, poor oral hygiene, human papillomavirus (HPV), Epstein-Barr virus, and chronic candidiasis.1-6
Clinical Presentation
Because oral leukoplakia is a painless and generally asymptomatic condition, it often goes unnoticed unless careful inspection is taken by both patient and primary care provider. Upon inspection of the tongue, buccal mucosa, and floor of the mouth, a white patch will be seen that can vary in appearance. Leukoplakia presents in 3 different forms:
- Homogenous: typically presents as a white, thin plaque that is even in color and smooth or wrinkled in appearance and consistent throughout; this form has a lower risk for malignancy1,2
- Nonhomogenous: presents as a white lesion that is typically irregularly shaped with inconsistencies throughout and may show erythema; this form has a higher risk for malignancy1,2
- Proliferative verrucous: presents as a white, hyperkeratotic, wart-like lesion. This form is aggressive, associated with HPV and EBV, and has a high malignancy rate.6
Diagnosis of Oral Leukoplakia
The differential diagnosis should include oral candidiasis, oral lichen planus, oral erythroplakia, oral hairy leukoplakia, or nicotine stomatitis in smokers.2,6,11 Scraping the lesion is important during the initial workup. In oral leukoplakia, the lesion will remain intact, while in oral candidiasis the plaque will be removed upon scraping and will bleed.2,11 A potassium hydroxide smear should be performed to identify Candida albicans as the causative agent. HIV testing may also be considered if suspicious of hairy leukoplakia or oral candidiasis without any known risk factors such as recent use of antibiotics, inhaled corticosteroids without use of a spacer, or chemotherapy.12 Other tests to consider for the workup of a white oral lesion may include hepatitis C antigen and antibody, which are linked to oral lichen planus and dermal lesions on the body.13 Lastly, because HPV is a risk factor for leukoplakia cases, clinicians should consider HPV testing.
Once other causes of white plaques have been ruled out through noninvasive testing, biopsy can be performed for a definitive diagnosis.2 Biopsy options include incisional and punch biopsy, which are performed to examine for histologic changes to the cells in the lesion. An excisional biopsy, which includes removal of the whole lesion, may be considered as well if the plaque is small in size. For larger lesions, an incisional biopsy is used and includes adjacent healthy tissue. Common histologic changes that are associated with oral leukoplakia seen on biopsy include loss of polarity of basal cells, nuclear hyperchromatism, nuclear pleomorphism, keratinization of cells, loss of intercellular adherence, increased nuclear-cytoplasmic ratio, and irregular epithelial stratification.2,7 A biopsy can also determine if the white lesion is benign, dysplastic, or has transformed to in situ or invasive carcinoma. The histologic results and lesion staging also play a key role in assessing which treatment options are most appropriate for the patient.
Although brush biopsy, in which a brush is swept along the lesion to collect cells, may be performed before surgical biopsy, evidence of the accuracy of this technique is mixed.2,8-10
Treatment Options for Oral Leukoplakia
As a premalignant condition with a high likelihood of progression to squamous cell carcinoma, immediate treatment of oral leukoplakia is encouraged. The main goal of treatment is to prevent further dysplasia and excision of moderate to severe dysplasia or carcinoma. A key aspect of management is removal of the primary source of irritation.2 This most commonly involves tobacco cessation as well as avoiding alcohol to help prevent further dysplastic changes to the tissue.
Surgical management is the gold standard for treatment of oral leukoplakia, however, patients may inquire about conservative options. Retinoids, beta carotene, and isotretinoin have been used to help reduce lesion size; however, clinical evidence on these treatments is very limited.3,14
A Cochrane review found that clinical improvement of lesions was achieved in studies using systemic vitamin A or retinoids (2 studies) and systemic beta carotene or carotenoids (1 study).14 Meta-analysis of 2 studies showed no benefit of beta carotene or carotenoids on the risk for cancer development.14,15 The review also reported lesion recurrence in 56% of patients treated with vitamin A and 54% of those given carotenoid management.14 Although the success and recurrence rates are variable and limited in study, it is important to discuss medical management as an alternative to surgical procedures. Clinicians should continue to discuss these options to provide patient-centered care, allowing patients to make informed decisions.
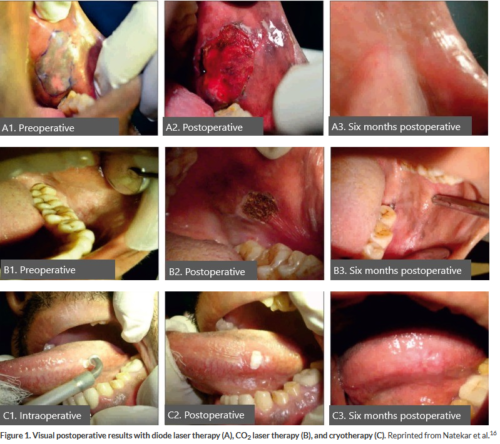
Surgical options for the treatment of oral leukoplakia include cryotherapy, diode laser therapy, CO2 laser therapy, and scalpel excision.2 Cryotherapy is a safe and simple selection in which a cryoprobe at a temperature of approximately -65 ºC is applied to the lesion to freeze and destroy the affected tissue.6 Diode laser therapy is another surgical option that involves the use of a concentrated beam of light for precise excision of the lesion with simultaneous hemostasis. Treatment with CO2 laser therapy allows for precise control of margins, minimal bleeding, decreased surgical time, minimal postoperative complications, and minimal scarring.15 Full excision involves the use of a scalpel to completely remove the lesioned area. Although a viable option, traditional scalpel removal is associated with increased intraoperative bleeding, postoperative infection, and possibly disfiguring results.
Comparison of CO2 Laser Therapy vs Cryotherapy
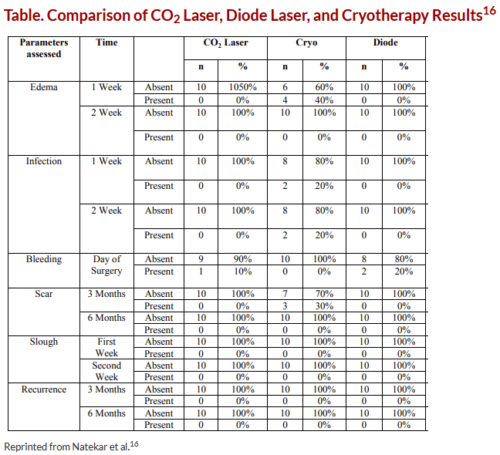
In a 2017 study, Natekar and colleagues compared outcomes of CO2 laser therapy, diode laser therapy, and cryotherapy in the treatment of oral leukoplakia (Figure).16 All 3 treatments were effective in the treatment of oral leukoplakia; however, both CO2 and diode laser therapies were associated with significantly lower rates of pain, hemorrhage, edema, and infection compared with cryotherapy (Table; P >.05). None of the patients in the laser therapy and cryotherapy groups developed scar formation compared with 30% of the cryosurgery group at 3 months; by 6 months all scars had resolved. None of the patients experienced recurrence at 6-month follow-up. While all treatment options should be considered for oral leukoplakia, postoperative risk and benefits should be discussed with patients to aid in the informed decision-making process.
Treatment Challenges With CO2 Laser Therapy
One challenge facing clinicians is deciding which patients may benefit from CO2 laser therapy. In a case study published by Pedrosa and colleagues, 59 patients with oral leukoplakia underwent 3 treatments of CO2 laser therapy, with 4-week intervals between each session.17 Results from this study show that 94% of patients with no or low-grade dysplasia were disease-free at 6-month follow-up. Lesion relapse at the treatment site occurred in 24 people (41%) at approximately 17 months postoperatively, and 25% of those patients (n=6) experienced transformation to squamous cell carcinoma.17
Initial histology from those patients who transformed to SCC revealed moderate- to high-grade dysplasia prior to CO2 laser treatment. CO2 laser therapy can still be used for moderate- to high-grade dysplasia; however, the increased recurrence rate should be discussed with the patient as well as the possible need for additional treatment with the more aggressive modality of traditional scalpel excision.
Another challenge of the CO2 laser, as well as for all therapies, is providing full application and treatment to all dysplastic cells. Due to the possibility of incomplete treatment, an extension of margins should be performed at least 3 mm beyond the lesion.15 The extension of laser therapy can further ensure destruction of dysplastic cells surrounding the lesion and aid in minimizing recurrence rates.
Prevention of Oral Leukoplakia
Primary care providers can aid in the primary prevention of oral leukoplakia by routinely screening their patients for this condition; risk factors include the use of tobacco products and excess alcohol consumption. Additionally, while taking a patient’s history, providers should inquire about oral hygiene routine, regularity of dental care visits, and any barriers that may affect the ability to receive oral health care. The survey by the American Dental Association showed that only 58% of respondents reported visiting a dentist at least once per year.18
If a patient is unable to visit a dentist biannually or maintain proper oral hygiene, the primary care provider can begin dental patient education and refer the patient to alternative and more affordable care options, such as public dental clinics or dental schools that can aid in oral health for minimal fees. Patient education should be offered to all individuals regarding the development of oral cancers caused by tobacco products and heavy alcohol use. If applicable, the provider can develop a treatment plan, counsel, or refer the patient for nicotine and or alcohol dependence.
Secondary prevention refers to monitoring the disease. Patients diagnosed with oral leukoplakia require frequent oral cavity examinations to identify any possible changes; especially because clinically lesions are asymptomatic. Thus, it is essential for primary care clinicians to be proactive and assess the oral cavity in all patients, especially those with risk factors, to promptly diagnose conditions such as oral leukoplakia. The earlier leukoplakia is diagnosed, the sooner treatment options can be discussed and performed.
Conclusion
Leukoplakia is one of the most common oral disorders that may progress to carcinoma. When discussing treatment options with patients, it is essential to discuss the risks and benefits associated with each possible procedure. It is also critical for clinicians to emphasize the importance of health care maintenance, including cessation of smoking and frequent follow-up with primary care providers and dentists, even after treatment, for inspection of new lesions or possible recurrence.Conclusion
Authors:
Christina Alvarez, PA-C, is an Internal Medicine and Pediatrics Physician Assistant and graduate from the St. John’s University Class of 2020 Physician Assistant Program in Queens, New York; Corinne I. Alois, MS, PA-C, is an assistant professor at St. John’s University Physician Assistant Program; Louise Lee, EdD, MHA, PA-C, is an associate professor and director of PA studies at St. John’s University.
References
1. Vail M, Robinson S, Condon H. Recognition of oral potentially malignant disorders and transformation to oral cancer. JAAPA. 2020;33(11):14-18. doi:10.1097/01.JAA.0000718268.52634.59
2. Mohammed F, Fairozekhan AT. Oral leukoplakia. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2021 Jan-. Updated July 27, 2021. https://www.ncbi.nlm.nih.gov/books/NBK442013/
3. Guimarães LM, Diniz MG, Rogatto SR, Gomez RS, Gomes CC. The genetic basis of oral leukoplakia and its key role in understanding oral carcinogenesis. J Oral Pathol Med. 2021 Aug;50(7):632-638. doi: 10.1111/jop.13140.
4. Macigo FG, Gathece LW, Guthua SW, Njeru EK, Wagaiyu EG, Mulli TK. Oral hygiene practices and risk of oral leukoplakia. East Afr Med J. 2006;83(4):73-8. doi:10.4314/eamj.v83i4.9419
5. Shang Q, Peng J, Zhou Y, Chen Q, Xu H. Association of human papillomavirus with oral lichen planus and oral leukoplakia: a meta-analysis. J Evid Based Dent Pract. 2020;20(4):101485. doi:10.1016/j.jebdp.2020.101485
6. Cleveland Clinic. Leukoplakia. Updated August 6, 2020. Accessed August 25, 2021. https://my.clevelandclinic.org/health/diseases/17655-leukoplakia
7. Parlatescu I, Gheorghe C, Coculescu E, Tovaru S. Oral leukoplakia – an update. Maedica (Bucur). 2014;9(1):88-93.
8. Kujan O, Idrees M, Anand N, Soh B, Wong E, Farah CS. Efficacy of oral brush cytology cell block immunocytochemistry in the diagnosis of oral leukoplakia and oral squamous cell carcinoma. J Oral Pathol Med. 2021;50(5):451-458. doi: 10.1111/jop.13153
9. Alsarraf A, Kujan O, Farah CS. Liquid-based oral brush cytology in the diagnosis of oral leukoplakia using a modified Bethesda Cytology system. J Oral Pathol Med. 2018;47(9):887-894. doi:10.1111/jop.12759
10. Reddy SG, Kanala S, Chigurupati A, Kumar SR, Poosarla CS, Reddy BV. The sensitivity and specificity of computerized brush biopsy and scalpel biopsy in diagnosing oral premalignant lesions: A comparative study. J Oral Maxillofac Pathol. 2012;16(3):349-353. doi:10.4103/0973-029X.102482
11. Mortazavi H, Safi Y, Baharvand M, Jafari S, Anbari F, Rahmani S. Oral white lesions: an updated clinical diagnostic decision tree. Dent J (Basel). 2019;7(1):15. doi:10.3390/dj7010015
12. Rathee M, Jain P. Hairy leukoplakia. In: StatPearls [Internet]. StatPearls Publishing; 2021 Jan-. Updated April 17, 2021. Accessed August 25, 2021. https://www.ncbi.nlm.nih.gov/books/NBK554591/
13. Arnold DL, Krishnamurthy K. Lichen planus. In: StatPearls [Internet]. StatPearls Publishing; 2021 Jan-. Updated August 11, 2021. Accessed August 25, 2021. Available from: https://www.ncbi.nlm.nih.gov/books/NBK526126/
14. Lodi G, Franchini R, Warnakulasuriya S, et al. Interventions for treating oral leukoplakia to prevent oral cancer. Cochrane Database Syst Rev. 2016;7(7):CD001829. doi:10.1002/14651858.CD001829.pub4
15. Romeo U, Mohsen M, Palaia G, Bellisario A, Del Vecchio A, Tenore G. CO2 laser ablation of oral leukoplakia: with or without extension of margins? Clin Ter. 2020;171(3):e209-e215. doi:10.7417/CT.2020.2215
16. Natekar M, Raghuveer HP, Rayapati DK, et al. A comparative evaluation: Oral leukoplakia surgical management using diode laser, CO2 laser, and cryosurgery. J Clin Exp Dent. 2017;9(6):e779-e784. doi:10.4317/jced.53602
17. Pedrosa A, Santos A, Ferreira M, Araújo C, Barbosa R, Medeiros L. Is carbon dioxide laser vaporization a valuable tool in the management of oral leukoplakia? A survey at an oncology hospital. Lasers Med Sci. 2015;30(5):1629-30. doi:10.1007/s10103-014-1551-2
18. American Dental Association. Survey: more Americans want to visit the dentist. ADANews. March 21, 2018. Accessed August 25, 2021. https://www.ada.org/en/publications/ada-news/2018-archive/march/survey-more-americans-want-to-visit-the-dentist

Leave A Comment
You must be logged in to post a comment.